Sub-Acute Budd-Chiari Syndrome
DOI:
https://doi.org/10.60591/crspmi.572Keywords:
Budd-Chiari Syndrome, Janus Kinase 2/genetics, Myeloproliferative DisordersAbstract
Budd–Chiari syndrome (BCS) is a rare vascular liver disorder characterized by obstruction of the hepatic venous outflow tract that is frequently associated with underlying prothrombotic conditions. Myeloproliferative neoplasms (MPNs), particularly those harboring JAK2 mutations, represent a major etiological factor.
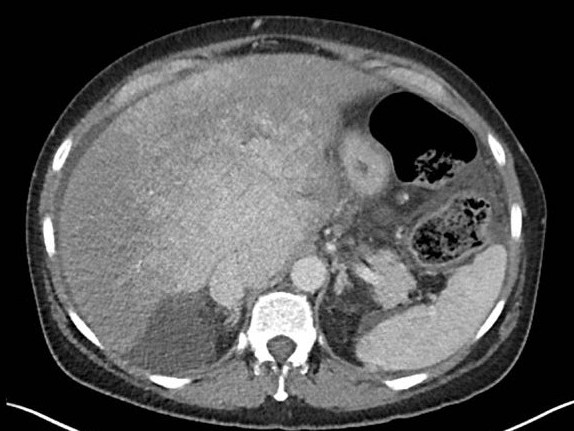
This case report describes a 53-year-old woman who presented with abdominal pain and ascites. Imaging revealed typical features of BCS, including hepatosplenomegaly, caudate lobe hypertrophy, heterogeneous liver parenchyma and prominent portosystemic collaterals. Hematological workup identified a JAK2 V617F-positive prefibrotic primary myelofibrosis as the underlying cause. Under medical therapy ascites resolved completely. Three years after diagnosis, the patient developed blue toe syndrome as a thromboembolic complication, prompting initiation of cytoreductive therapy with hydroxycarbamide and antiaggregant treatment with aspirin.
This case highlights the importance of early diagnosis and comprehensive evaluation for underlying prothrombotic conditions in patients with BCS. Routine screening for MPNs, including JAK2 mutations, is essential. Prompt anticoagulation and an individualized, multidisciplinary management approach are crucial for improving patient outcomes.
Downloads
References
1. Li Y, De Stefano V, Li H, Zheng K, Bai Z, Guo X, et al. Epidemiology of Budd-Chiari syndrome: A systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2019;43:468-74. doi: 10.1016/j.clinre.2018.10.014.
2. Janssen HL, Garcia-Pagan JC, Elias E, Mentha G, Hadengue A, Valla DC, et al. Budd-Chiari syndrome: a review by an expert panel. J Hepatol. 2003;38:364-71. doi: 10.1016/s0168-8278(02)00434-8.
3. Menon KV, Shah V, Kamath PS. The Budd-Chiari syndrome. N Engl J Med. 2004;350:578-85. doi: 10.1056/NEJMra020282.
4. Garcia-Pagán JC, Valla DC. Primary Budd-Chiari Syndrome. N Engl J Med. 2023 ;388:1307-16. doi: 10.1056/NEJMra2207738.
5. Darwish Murad S, Plessier A, Hernandez-Guerra M, Fabris F, Eapen CE, Bahr MJ, et al. Etiology, management, and outcome of the Budd-Chiari syndrome. Ann Intern Med. 2009;151:167-75. doi: 10.7326/0003-4819-151-3- 200908040-00004.
6. Zeitoun G, Escolano S, Hadengue A, Azar N, Younsi El, Mallet A, et al. Outcome of Budd-Chiari syndrome: a multivariate analysis of factors related to survival including surgical portosystemic shunting. Hepatology.1999;30:84–89
7. De Gottardi A, Trebicka J, Klinger C, Plessier A, Seijo S, Terziroli B, et al. Antithrombotic treatment with direct-acting oral anticoagulants in patients with splanchnic vein thrombosis and cirrhosis. Liver Int. 2017; 37:694–699.
8. Thapa SB, Souza GR, Paravathaneni M, Cohen S, Mohammed T, Laber DA. Direct oral anticoagulants in Budd–Chiari syndrome. Eur J Haematol. 2025;114:566–72.
9. European Association for the Study of the Liver. Electronic address: easloffice@ easloffice.eu; European Association for the Study of the Liver. EASL Clinical Practice Guidelines on vascular diseases of the liver. J Hepatol. 2026;84:399-456. doi: 10.1016/j.jhep.2025.08.001.
10. De Stefano V, Vannucchi AM, Ruggeri M, Cervantes F, Alvarez-Larrán A, Iurlo A, et al. Splanchnic vein thrombosis in myeloproliferative neoplasms: risk factors for recurrences in a cohort of 181 patients. Blood Cancer J. 2016;6:e493. doi: 10.1038/bcj.2016.103.
11. Guglielmelli P, Pacilli A, Rotunno G, Rumi E, Rosti V, Delaini F, et al. Presentation and outcome of patients with 2016 WHO diagnosis of prefibrotic and overt primary myelofibrosis. Blood. 2017;129:3227-36. doi: 10.1182/blood- 2017-01-761999.
12. McLornan DP, Psaila B, Ewing J, Innes A, Arami S, Brady J, et al. The management of myelofibrosis: A British Society for Haematology Guideline. Br J Haematol. 2024;204:136-50. doi: 10.1111/bjh.19186.
Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2026 Andréa de Gottardi, Silke Klapdor

This work is licensed under a Creative Commons Attribution 4.0 International License.






